September 13, 2022 Industry news
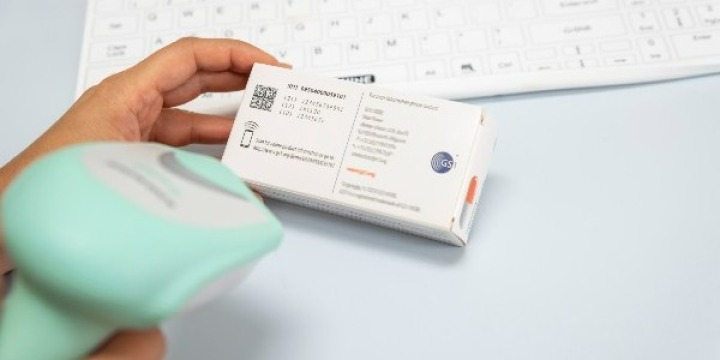
August 2022 marked a significant milestone in patient safety with the scan of the first serialised vaccine in Africa.
For several years, multi-national partners including UNICEF, Gavi, The Bill & Melinda Gates Foundation, the Global Fund, USAID, national regulatory authorities in Nigeria and Rwanda, Vital Wave, and the World Bank have been collaborating under a partnership known as The Verification and Traceability Initiative (VTI). The VTI was established to “increase global coordination and response against falsified medical products, especially in low- and middle-income countries”.

The partnership was formed to improve the traceability and authentication of medicinal products on the global market and the launch of the Traceability and Verification System (TRVST) has made this shared vision a reality.
TRVST, serves as a global repository that holds product-specific information submitted by the legal manufacturers of medical products. This includes details such as the GS1 Global Trade Item Number (GTIN), the unique product identifier; batch and serial number, and expiry dates.
Powered by GS1 standards
Built by GS1 UK-Approved partner SolidSoft Reply, the TRVST database allows products to be matched with, and checked against, the corresponding repository information before it is used. Each medicinal product is given its own unique identifier, allowing it to be authenticated and traced throughout the supply chain.
This unique identifier used by the TRVST is the GS1 GTIN. Each unique GTIN is licenced to the legal manufacturer of the item and encoded into a barcode (often a 2D DataMatrix) on the product’s packaging. Once at the point of care, the barcode can then be scanned before it is administered to a patient to ensure it is authentic.
UNICEF and VTI partners have been working to expand the TRVST by enhancing its verification and traceability capabilities. In July 2022, Rwanda and Nigeria became the first countries in Africa to authenticate lifesaving vaccines using GS1 standards.

The GS1 UK partner team has worked closely with SolidSoft Reply to approve their product at the pre-tender stage and collaboration will continue, supported by our colleagues at GS1 Global Office, as the rollout continues.
Read the full press release below.
Scanning to save lives
Falsified, substandard, and counterfeit medicines continue to threaten the safety of patients across the world and, according to the World Health Organisation (WHO) result in hundreds of thousands of deaths each year. The complexities of the pharmaceutical supply chain with its many touchpoints, often leaves the chain of custody at risk.

Although this is very much a global challenge, countries in which access to medicines is more restricted are at the greatest risk. In 2017, a WHO report anticipated that as many as 1 in 10 medicines available in low- and middle-income developing countries are substandard or falsified, costing “around US$200 billion globally and US$30 billion each year”.
However, it is the human cost that bears the biggest impact. “Counterfeit medicines are estimated to have caused up to 169,000 deaths of children under five suffering from pneumonia and up to 116,000 deaths from falsified medicines to treat malaria” each year.
Serialisation literally saves lives. It is essential that medical companies and healthcare provides can maintain, and have trust, in the integrity of supply chains if we are to prevent further harm to patients.
As World Patient Safety Day approaches, this news marks just the latest victory in the global fight against counterfeit medicines.