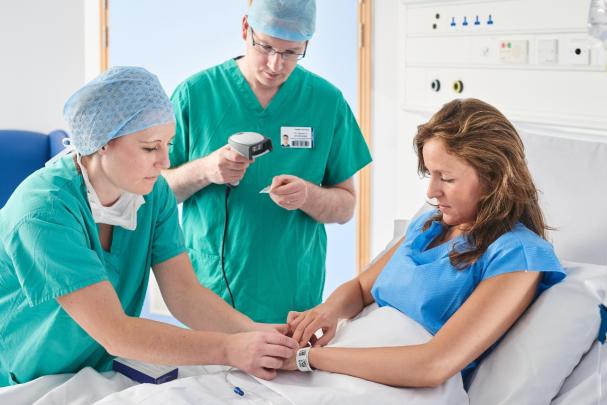
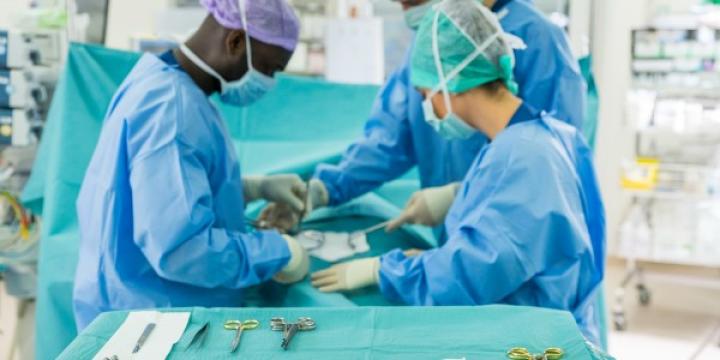
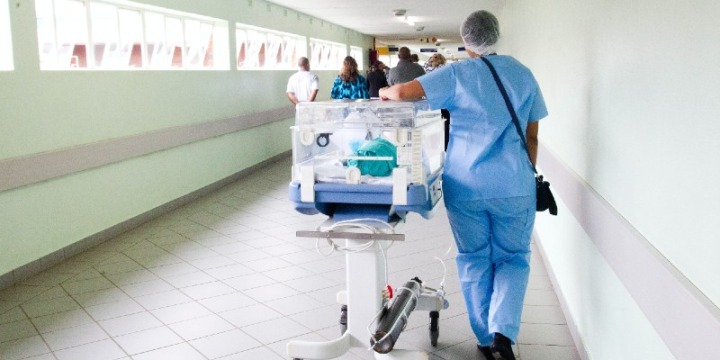
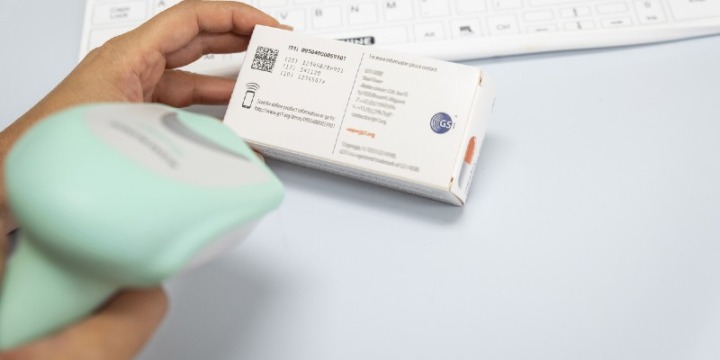
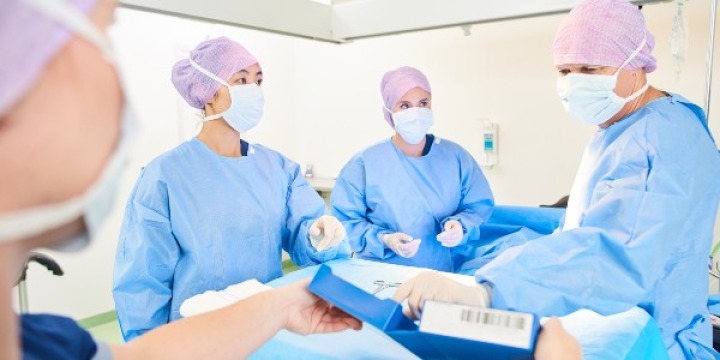
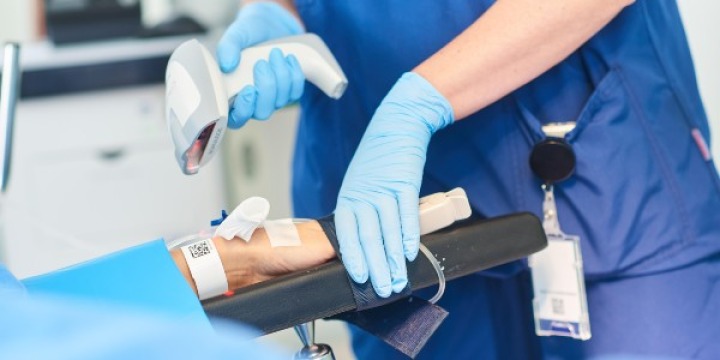
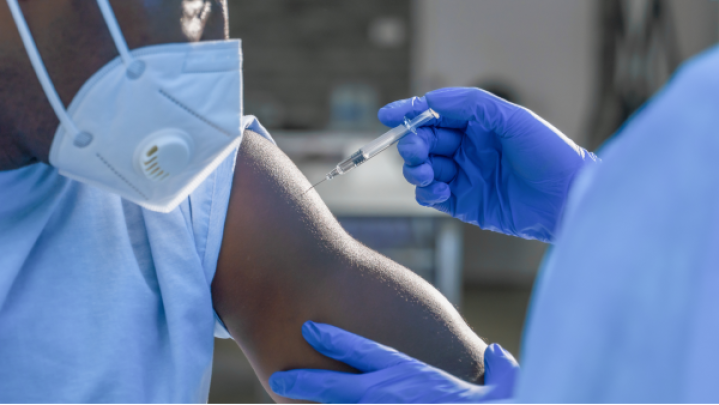
Here you will find information on how to adopt GS1 standards across the three core enablers (people, products and places), how to apply these to specific use cases such as surgical instrument traceability, and details on supporting regulatory requirements.
Take a look at our detailed guidelines for expert advice to support you along the way.